 Check out the article regarding the polarity of NH3. Memorize it ASAP!Īmmonia is also one of the compounds having the same geometrical structure ie trigonal pyramidal. This chart is very important hence, it helps to answer such questions. So, the molecular geometry for ph3 is Trigonal Pyramidal. When a steric number four comes along with a lone pair we have a Trigonal Pyramidal forming just as shown in the VSEPR table below. If we start off counting the number of groups or the number of atoms attached to the phosphorus, we get one, two, three, and the lone pair which is four that gives us the term, what’s called the steric number which is four in this case. So, in the lewis structure of PH3 above, it’s visible that we have three hydrogens along with a lone pair that is formed on the top of it. Let’s take a look at the molecular geometry of ph3 (phosphorus tri-hydrate) closely! But in NCl3, Nitrogen, and Chlorine, both have the same electronegativity of 3.0, so the molecule NCl3 is Non-Polar.įor more understanding, check out the article for the polarity of NCl3.Īlthough, both compounds are Trigonal Pyramidal! Hence, PH3 has an electronegativity difference forming polar bonds. Let’s clear the mist behind this, the molecule NCl3 has the same structural behavior as PH3 but PH3 has a negatively charged lone pair on along with 3 positive hydrogens present in the lewis structure giving rise to unequal charges in the compound which we’ve discussed earlier! Dipole moment is the value of the measurement of the polarity extent of a compound. Hence, the Phosphine compound is a polar molecule. This lone pair of electrons becomes a negative region and the hydrogen forms a positive region as each hydrogen’s electrons will most likely occupy the bonds occurring between the hydrogen and phosphorus excluding the hydrogen’s positively exposed nucleus.Īnd as a rule of polarity, any molecule with a positive along with a negative region will be considered polar. Therefore, a lone pair of electrons is left behind. Here, three hydrogens give 3 electrons to the central atom and thus, satisfy the octet rule for (P). In the PH3 molecule, the Phosphorus atom has 5 valance electrons. When the charge distribution is equal between the atoms of a diatomic molecule, or when the polar bonds in a large molecule are able to cancel out each other, a nonpolar molecule is formed. A compound becomes Polar when an electronegativity difference is found between the bonded atoms.ģ. Polarity is the electric charge distribution around atoms, molecules, or chemical groups.Ģ. 
We can answer this based on the following terms:ġ. Its IUPAC name is phosphane and it is slightly soluble in water and hence is considered to be insoluble which we will discuss later!īefore we dive deeper, let’s recall a few terms.Ĭonclusion How to differentiate a polar or a non-polar molecule? It is generally classed as a pnictogen hydride in chemistry. 
Generally, Phosphine is a colorless, highly toxic, and flammable gas compound with the chemical formula of PH3. PH3 (Phosphine) is also polar because of the rule that all polar molecules must contain polar bonds which are formed due to a difference in electronegativity found between the bonded atoms of the chemical compound. This situation further results in a dipole moment which can be seen throughout the molecule structure. 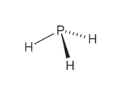
So, is PH3 Polar or Nonpolar? PH3 is a polar compound due to the presence of a lone pair of electrons with an electron-electron repulsion which gives rise to an overall “bent” structure. Now that you know, a brief history of this toxic compound, let’s come back to its polarity!! Its pure form is odorless, and other forms have an unpleasant odor like a rotten fish, or garlic, due to the presence of substituted Phosphine and Diphosphane. Hence, the compound is crushed to the core and gains the ability to heat a talk to a debate. Phosphine or AKA Phosphorus Tri-hydrate (PH3) is the most misunderstood chemical compound in chemistry and the reason is it’s a polar molecule with non-polar bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
